How are objects made from natural resources created?
On the last page I included some things that showed how they are created. On this page, I am going to focus on how they are created, not their materials.

Here is how bike frames are made. Seemless frame tubes are created from steel blocks that are soild, punturcted, and "drawn" into tubes through sveral stages. This is one of the ways to make the frames. The alterantive is seemed tubes, which are manufactured by drawing flat steel srio stock, wrappunngit in a tube, and welding it together along the side of the tube.
The tubes are assembled by welding by the machine. Composties can be joined together by strong glue or plastic binders. The parts are mostly made by the machiine and can be connected to the frame by the machine or hand. The final adjustments are done by talented bicycle builders.
The tubes are assembled by welding by the machine. Composties can be joined together by strong glue or plastic binders. The parts are mostly made by the machiine and can be connected to the frame by the machine or hand. The final adjustments are done by talented bicycle builders.
To tailor the tubes, first the metal is softened by heating. It is hallowed out to form "hallows". Those are reheated. Then they are put in acid to remove scale, and a substance (such as oil or grease) is applyed to make it smoother.
Next, the hallows are cut, measured, and prescion metered to the correct dimensions. Then, they are fitted over a rod and attached with a bench. To get the right gauge, the hallows go through a process called cold drawing. That is where they pass through dies which strech them into thinner and longer tubes.
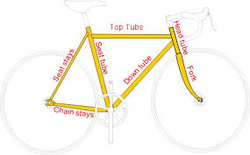
Like I said earlier, the tubes can be added into a frame by hand or machine. Frames can be brazed (basically welding at a temperature around 1600 degrees Farenheight or lower), welded or glued. This can be done without lugs. Lugs are metal sleeves joining two or more tubes at a joint. Gas burners are prepared around lugs, which afre being heated for brazing. The brazer filler is often bronze or silver, which melt at lower temperatures than the tubes being joined. As the bronze or silver melts, the filler is applied. It flows around the joint, sealing it.
The assemlbed frames are placed into jogs and checked for correct allignment. Any adjustments nessesary are made now. The extra flux and brazing materials are removed. Once the metal has cooled, the last corrections are made. Finallly, the frames are painted. This is not just for a completed look, but to protect the frames. The frame is first made ready with an undercoat and then painted with a colored enamel. Paint can be added by hand-spraying or by passing the frames through automatic electrostatic spraying rooms. The negatively charged frames attract the positively charged paint spray as the frames rotate for full coverage. Finally, transfers and lacquer are added to the frame.
Now for aluminum foil. Aluminum foil is made in two major stages which are:
- Aluminum oxide (alumina) is refined from bauxite in a several-step caustic soda/high temperature process.
- To seperate it into aluminum and oxygen, the alumina is electronically reduced in molten cryolite.

Shortly, to get the alumina, the bauxite ore is broken up and screened when necessary to get rid of stone and similar large impurities. Next, it is combined with a caustic liquor in slurry tanks and pumped to steam-heated digesters, where added caustic dissolves the alumina from the bauxite. Were the red mud collectts at tthr bottom, leaving the alumina on top, is where the entire mix is transfered to: in the setting tanks. It now passes through filters to remove the last of the unwanted mud, and is held in large tank, with some very fine particles still suspended as the liquor overflows the setting tank. the liquor is seeded with a fine alumina hydrate and air-agitated to precipitate crystals of hydrated aluminum oxide when cool.
All remaining water is driven from the pure aluminum oxide by being washed, drained, and transferred to high temperature rotary kilns. the alumina is stored for delivery to aluminum reduction plants when cool. The alumina is changed into aluminum at the reduction plant. Normally, this is completed in a carbon-lined electrolytic cell in which the electrolyte is molten cryolite. This is practically the hall process. The current flows inbetween a carbon anode and the pot lining. As alumina is combined to the bath, it is divided by electrolysis into oxygen and aluminum. The liberated oxygen combineis added with the carbon of the anode and is carried away as carbon dioxide gas. The molten aluminum is not lighter than the electrolytic solution and stays to the bottom of the reduction cell, from which it is siphoned at appropriate intervals.
The aluminum reduction process operates at low voltage and high amperage. A series of these electrolytic cells or pots joined together by biig aluminum electrical bus bars, is named a potline. The molten aluminum is siphoned off into crucibles which are moved by overhead cranes and removed into furnaces, where other elements are combined to make the desired alloy. The metal is then poured into vertical, direct chill casting devices to create rolling ingot. Ingot is reduced to re-roll stock gauges on the regular sheet and plate mills, and finally to foil gauges in specialized foil rolling mills. Re-roll stock also is made directly from molten metal. Two pounds of alumina, gathered from four pounds of bauxite, are needed to create one pound of aluminum. Also, 0.6 pound carbon and small amounts of other materials, are consumed in the process. When commercial production of aluminum started shortly before the turn of the century, 12 kilowatt hours (Kwh) of electrical energy were needed to make one pound of aluminum. By the early 1950's, process refinements had reduced the power consumption to about 9. In the late 1970's, the average is less than 8 Kwh/lb, with a few newer plants having gotten a 6.5 Kwh/lb energy use. And one pilot plant, now using a new aluminum smelting process, has consumed less than 5 Kwh/lb average during its first nine months of normal operation. The aluminum industry is still employing energy conservation practices and research at all levels.
Now the foil is techically made now.

